Author: Dr Gerald Liew

Colour fundus photographs showing widespread retinal pigment epithelium and choroidal atrophy. There is mild arteriolar attenuation. Some pigment deposition is visible in the right macula.
A 46 year-old Caucasian male presented with
increasing difficulty seeing in the dark.
Case History
A 46-year-old Caucasian male described increasing difficulty seeing and driving in the dark over the last 5 years. When questioned he admitted to having had some trouble with night vision since his early 20s. When he went dancing in night clubs, he was often unsure who he was dancing with, but assumed other people had the same experience. Central vision remained unaffected. There was no significant past ocular history. Past medical history was unremarkable other than mild hypertension. He denied use of any medications other than anti-hypertensive medications. There was a vague family history of a male relative in the United Kingdom who had been diagnosed with a serious eye disease several years ago.
On examination his uncorrected visual acuities were 6/9 in the right eye (OD) and 6/6 in the left eye (OS). Intraocular pressures were 14mmHg OD and 16mmHg OS. Anterior segment examination was unremarkable with clear media in both eyes. He had widespread areas of generalised retinal atrophy in both eyes in a symmetrical distribution with some retinal pigment deposition OD. There was mild arteriolar attenuation in both eyes (Figure 1). Optic disc appearances were unremarkable.
What is your diagnosis?
Differential diagnosis
The differential diagnosis of nyctalopia with generalised retinal atrophy includes inherited conditions such as retinitis pigmentosa, cone rod dystrophy, Bietti crystalline dystrophy, choroideremia, gyrate atrophy, other rarer retinal dystrophies; and acquired causes such as vitamin A deficiency.
Additional Investigations
Fundus autofluorescence imaging highlights the widespread retinal atrophy with lighter regions signifying areas of residual central retina (Figure 2). The edges of the atrophy were well defined ‘scalloped’ edges, consistent with disease affecting the retinal pigment epithelium.1 Optical coherence tomography (OCT) of the macula showed only a small area of residual outer retina (Figure 3). The patient contacted his relatives in the United Kingdom and found he had an older male cousin who had recently been diagnosed with an inherited retinal dystrophy. The family pedigree is consistent with X-linked inheritance (Figure 4). His maternal grandfather died young and little history was available regarding his eyesight.

Fundus autofluorescence showing marked, well defined ‘scalloped’ areas of retinal pigment epithelium loss (dark regions).
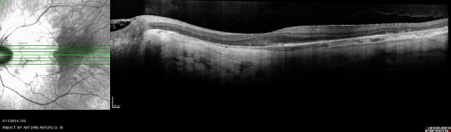
OCT of the left macula showing loss of retinal pigment epithelium and underlying choroid with only a small island of residual outer retina at the fovea.

Pedigree showing the proband (arrow) and an affected male cousin. The pattern is consistent with X-linked inheritance.
Diagnosis
The clinical signs, imaging and pedigree are consistent with choroideremia, an X-linked retinal dystrophy.
Clinical Course
The patient was informed of the likely diagnosis and contacted his cousin, who was found to have choroideremia confirmed on genetic testing in the United Kingdom.
Discussion
Choroideremia is an X-linked retinal dystrophy characterised by progressive degeneration of the retinal pigment epithelium, choroid and photoreceptors. The condition is an isolated ocular disease and is believed to occur with an incidence of 1 in 50-100,000 persons.2 Patients usually develop nyctalopia in the 1st or 2nd decade which progressively worsens. Electrophysiology testing can be useful in confirming the diagnosis and shows rod system dysfunction followed by cone system dysfunction late in the disease course.
The genetic defect has been mapped to mutations in the REP-1 (Rab Escort Protein) gene on chromosome Xq13-q22.2,3 The gene product is a protein that is involved in intracellular vesicular transport. The most common mutations are nonsense and frameshift mutations leading to complete loss of the gene product but other mutations such as splicing mutations and translocations are also described.2 Genetic testing is useful to confirm the diagnosis but is currently not widely available outside of research settings.
The patient was counselled that he likely inherited the genetic defect from his maternal grandfather and that his mother is a carrier. If he has children, all his future daughters will be carriers and none of his sons will be affected. His grandsons have a 50% risk of inheriting the condition. The patient had extremely restricted visual fields and voluntarily ceased driving. There is currently no known cure but early phase gene therapy trials in the United Kingdom have shown promising results in slowing progression of the condition and two out of six patients reported marked improvement in central vision.4 A diet rich in leafy green vegetables and carotenoids may help slow progression but this remains debatable as male patients often have normal levels of macular carotenoids.5
Take home points |
|
Want to subscribe to the Retina Associates Case of the Month? Click here!
References
1. Liew G, Halford S, Mackay DS et.al. Detailed phenotypic and genotypic characterization of bietti crystalline dystrophy. Ophthalmology 2014;121:1174-1184.
2. Coussa RG, Traboulsi EI. Choroideremia: a review of general findings and pathogenesis. Ophthalmic Genet 2012;33:57-65.
3. Preising M, Ayuso C. Rab escort protein 1 (REP1) in intracellular traffic: a functional and pathophysiological overview. Ophthalmic Genet 2004;25:101-110.
4. MacLaren RE, Groppe M, Barnard AR et.al. Retinal gene therapy in patients with choroideremia: initial findings from a phase 1/2 clinical trial. Lancet 2014;383:1129-1137.
5. Zhao DY, Wintch SW, Ermakov IV et.al. Resonance Raman measurement of macular carotenoids in retinal, choroidal, and macular dystrophies. Arch Ophthalmol 2003;121:967-972.
